Intelligent Closed-Loop Systems
Video of the demonstrator in the Neurobots project, 2016:
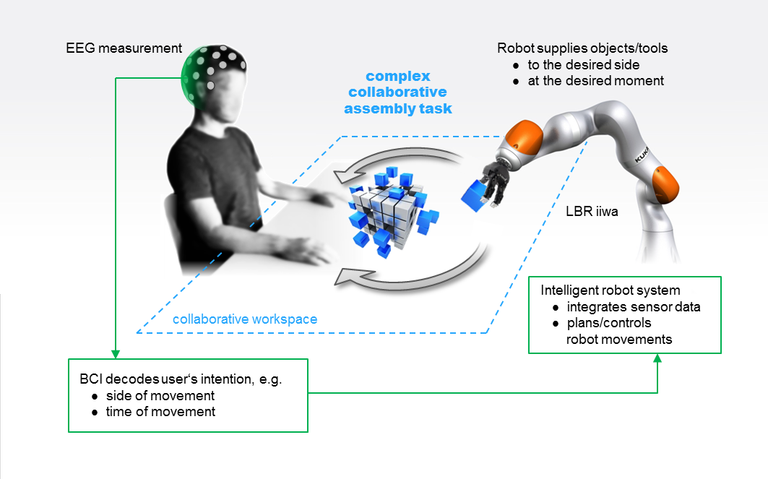
A common theme of our interdisciplinary research group at Translational Neurotechnology Lab is how to interpret the brain activity in order to control external devices, such as robots, or computer programs. Such devices, commonly called brain-computer interfaces (BCI) or neurally controlled robots (Neurobots, see video above), have the potential to greatly improve life quality of people with serious health issues, such as tetraplegia, or locked-in syndrome, by assisting them in their every-day tasks (see Fig. 1 for details of the BCI concept).
With our support from the BrainLink-BrainTools Cluster of Excellence awarded to University of Freiburg we have been working on solving this challenging problem. In a close cooperation with research groups of Prof. Wolfram Burgard (and their expertise in machine learning), Prof. Thomas Stieglitz (and their expertise in neuronal implants), and Prof. Andreas Schultze-Bonhage (and their expertise in intracranial signal measurements and analysis), we believe to bring these technologies to the people, who would need it most.
Figure 1: Concept of brain-computer interface (BCI). The problem of brain activity interpretation consists of several inter-related steps. First, it is necessary to measure the brain activity. Second, the brain activity must be processed and the relevant features decoded into user’s intention. Third, the decoded intention is then passed to an external device (a BCI or a robot), which can then more-or-less autonomously execute a series of commands to complete the task.
Our interdisciplinary research group is involved in the analysis of many different neuronal signals, ranging from activity of single neurons to large neuronal populations. We analyze various cognitive tasks (e.g., motor control, error processing, or speech production) and the corresponding brain activity, for instance, single unit activity (SUA), intracranial EEG, or scalp electroencephalography (EEG).
Somewhat historically, the main focus of our group has been on analysis of intracranial EEG (especially electrocorticography, or ECoG), mostly due to the close cooperation of our group with the Epilepsy Center at Freiburg University Medical Center. The intracranial EEG recordings are a very unique opportunity to study such a signal type, because – as the name suggests – it is measured under the skull. Such measurements would not be (due to ethical reasons) possible to healthy volunteers. However, there is a certain group of patients suffering from a pharmaco-resistent form of epilepsy, who undergo the electrode implantation during a presurgical monitoring to localize the epileptic foci. The patients are implanted with ECoG arrays (for example, 10 x 10 cm) or with brain-penetrating depth electrodes (so-called stereotactic EEG, or SEEG). During this period, it is possible to conduct several behavioral experiments with these patients, after receiving their informed consent and the approval by the university ethics committee. Therefore, we are very grateful to the patients, who – despite their extremely difficult life situation – make it possible to push the frontiers of science.
The intracranial EEG constitutes an interesting signal modality between the single neuron recordings on one hand (which are quite informative, but invasive and unstable in time) and the scalp EEG on the other hand (non-invasive and stable, but not so informative). Moreover, it is possible not only to read out the brain activity, but also to stimulate the brain directly. With the recent advent of micro-ECoG (µECoG; where the entire array is as big as a dime), the hopes are that the signal could be closer to the SUA as for the richness of its informational content, but with a superior stability in time (lasting for years to decades). We are also actively involved in the safety evaluation of the µECoG arrays (currently in sheep animal models), working in a close cooperation with Prof. Thomas Stieglitz group and the ALU spinoff company CorTec. If the µECoG arrays indeed turn out to be safe and ready-to-use in humans, as our preliminary research results suggest, there are many potential neurotechnological applications, for instance in brain-machine interface (BMI) research aiming at restoring lost motor and sensory functions of paralyzed people. It is our commitment and endeavor to bring this new neurotechnology to people and thus improve their quality of life.